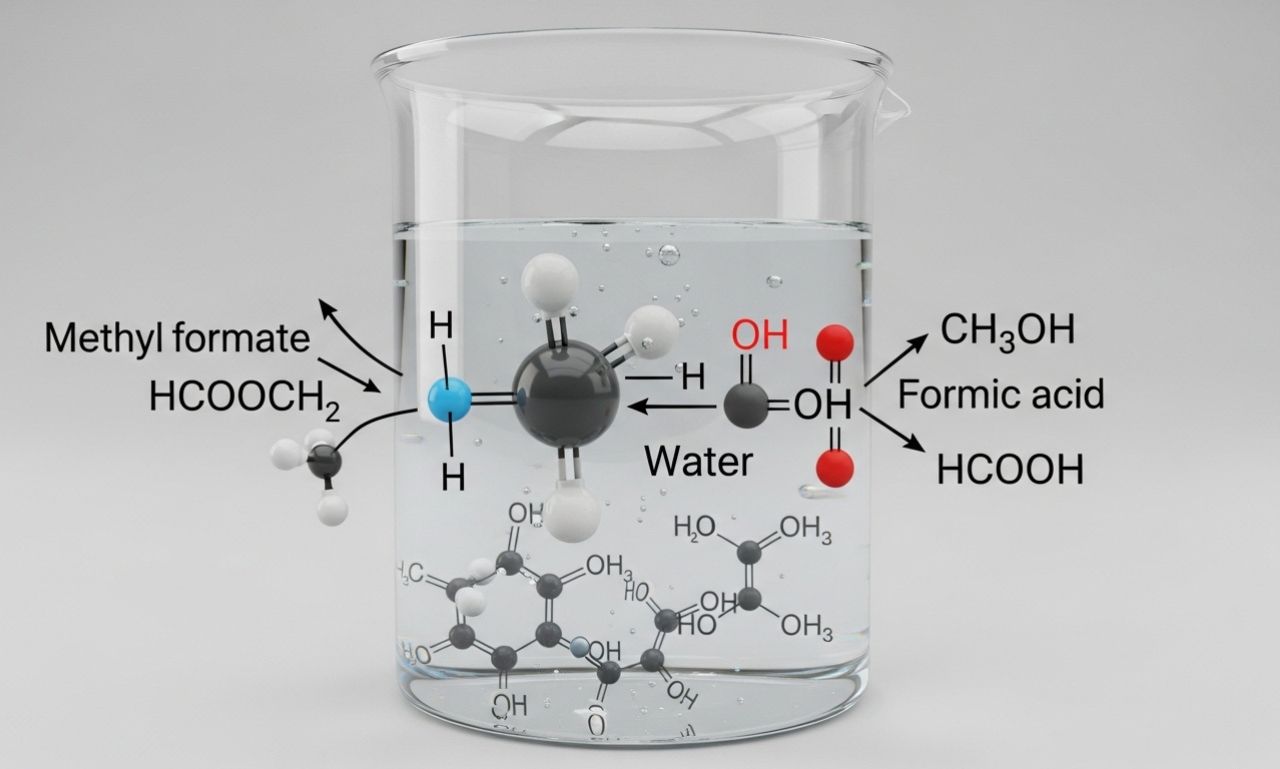
HCOOCH₂ + H₂O represents a simplified way of writing the hydrolysis reaction of an ester compound, commonly related to methyl formate (HCOOCH₃) reacting with water (H₂O). In organic chemistry, this type of reaction is known as ester hydrolysis, where an ester reacts with water to form an alcohol and a carboxylic acid.
This article explains the meaning, chemical reaction, mechanism, industrial importance, and applications of this process.
What is HCOOCH₂?
The formula HCOOCH₂ appears to be a shortened or incomplete representation of an ester. The correct and commonly known compound is:
-
HCOOCH₃ – Methyl formate
Methyl formate is an ester formed from:
-
Formic acid (HCOOH)
-
Methanol (CH₃OH)
Basic Properties of Methyl Formate
-
Chemical Formula: HCOOCH₃
-
Molecular Weight: 60.05 g/mol
-
Functional Group: Ester (-COO-)
-
Odor: Pleasant, ether-like smell
-
State: Colorless liquid
What Happens When HCOOCH₃ Reacts with H₂O?
When methyl formate reacts with water, a hydrolysis reaction occurs.
General Reaction:
HCOOCH3+H2O→HCOOH+CH3OHHCOOCH₃ + H₂O → HCOOH + CH₃OHHCOOCH3+H2O→HCOOH+CH3OH
Products Formed:
-
Formic Acid (HCOOH)
-
Methanol (CH₃OH)
This reaction can occur under:
-
Acidic conditions (acid-catalyzed hydrolysis)
-
Basic conditions (base-catalyzed hydrolysis, also called saponification)
Acid-Catalyzed Hydrolysis
In acidic conditions (usually using sulfuric acid as a catalyst), the ester reacts slowly with water.
Reaction Type:
Reversible reaction
Mechanism Steps:
-
Protonation of the carbonyl oxygen
-
Nucleophilic attack by water
-
Rearrangement and elimination
-
Formation of acid and alcohol
This process is part of the broader study of ester reactions in organic chemistry.
Base-Catalyzed Hydrolysis (Saponification)
In basic conditions (using NaOH or KOH), the reaction proceeds faster and is irreversible.
Reaction:
HCOOCH3+NaOH→HCOONa+CH3OHHCOOCH₃ + NaOH → HCOONa + CH₃OHHCOOCH3+NaOH→HCOONa+CH3OH
Products:
-
Sodium formate
-
Methanol
This method is commonly used in laboratory and industrial processes.
Industrial Importance of HCOOCH₂ + H₂O
The hydrolysis of methyl formate has several practical applications:
1. Production of Formic Acid
Formic acid is widely used in:
-
Leather processing
-
Textile industries
-
Agriculture (silage preservation)
2. Production of Methanol
Methanol is used in:
-
Fuel production
-
Chemical synthesis
-
Manufacturing plastics
Chemical Significance of HCOOCH₂ + H₂O
The reaction between HCOOCH₃ and H₂O demonstrates important organic chemistry principles:
-
Ester functional group behavior
-
Nucleophilic substitution reactions
-
Acid-base catalysis
-
Reaction equilibrium concepts
It is often studied in undergraduate organic chemistry courses as a classic example of ester hydrolysis.
Safety Considerations of HCOOCH₂ + H₂O
-
Methanol is toxic if ingested or inhaled.
-
Formic acid can cause burns and irritation.
-
Proper laboratory safety equipment should always be used.
Conclusion on HCOOCH₂ + H₂O
The expression HCOOCH₂ + H₂O refers to the hydrolysis of an ester, most accurately methyl formate (HCOOCH₃), in the presence of water. This reaction produces formic acid and methanol and plays a significant role in both laboratory studies and industrial chemistry.
Understanding this reaction helps students grasp fundamental concepts of ester chemistry, reaction mechanisms, and practical chemical manufacturing processes.
If you’d like, I can also provide a reaction mechanism diagram or a simplified explanation for exam preparation.